|
7/14/2023 0 Comments Ion bonding capacity measure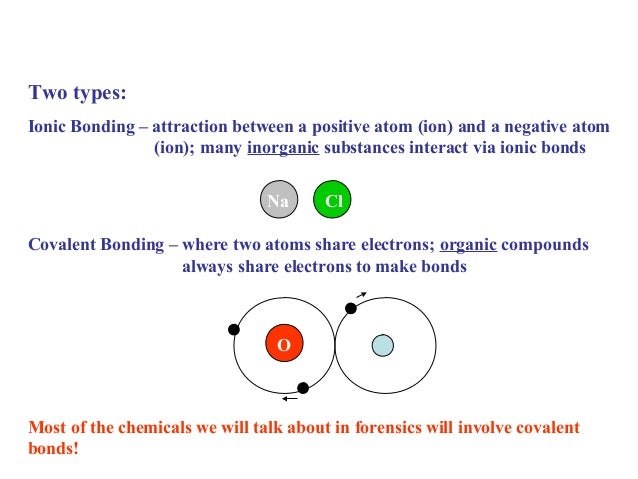
Stay tuned for my next article which will take a deeper dive into strategies for improving equity and how this can increase capacity. To answer more questions, you can email be at or call my direct line at (619) 937-0166. 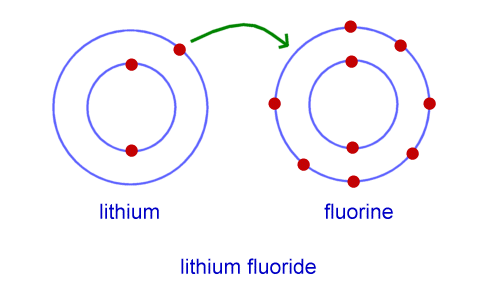
To find out what your bonding capacity looks like, request a quick capacity analysis and I will provide you with the information you need for your company. Determining what type of bonding capacity you can establish and/or deserve is a key part of the process. When reviewing the balance sheet and income statement, two important items that an underwriter will be reviewing are the contractor’s working capital and their equity. We took a deeper dive into working capital in a previous article, but simply put, working capital represents a contractor’s current assets minus current liabilities, and this measures how much a company has available to pay its current debts. Equity, or net worth on the balance sheet, is made up of retained earnings, common stock and additional paid in capital, and these numbers provide a measure of the long term liquidity of a company. Surety carriers take a hard look at this number because they want to ensure that there are sufficient reserves to complete the work that they have issued performance and payment bonds on.īuilding an effective bonding program can take time and requires collaboration with competent, trusted advisors. IEC is a key feature of anion-exchange membranes (AEM), as it determines the AEM’s ability to conduct the ions required to sustain the electrochemical reactions where they are utilized. Generally speaking, a contractor’s bonding capacity is comprised of single and aggregate limits, where the surety underwriter will approve performance and payment bonds for a job, up to the single limit. The aggregate limit is the cap that the surety carrier sets for how much total bond liability a contractor can have extended at one time. Having these caps is what makes it important for contractors to have an understanding of what information sureties use when determining how much capacity to offer. Underwriters will look at personal and business credit, industry experience, as well as personal financial wealth. Typically, though the most important item a surety underwriter will focus on is the company’s financials, specifically, their balance sheet and income statement. Ion-exchange capacity (IEC) is the measure of a material’s capability to displace ions formerly incorporated within its structure. So, what is bonding capacity, and what items determine the amount of capacity that a surety carrier is willing to offer? 
Ion-dipole interactions are electrostatic forces of attraction between an ion and a polar molecule. Dipole refers to the partial negative and positive charges on a molecule. Therefore they can conduct as their ions are free to move.For contractors that do a lot of bonded work, their bonding capacity is a critical element of their business. Capacity often determines which projects a company can and cannot pursue, so it is managed very closely. However, for contractors that are new to bonding or have not bonded previously but remain interested in performing bonded work, this is likely a foreign concept to them. An ion is a positively charged (cation) or negatively charged (anion) species. Ionic compounds dissolve in water easily, when they do this their lattice breaks up completely and water molecules surround the seperated ions. A cube of tightly packed alternate metal and non-metal ions. This is a regular repeating arrangement of metal and non-metal ions which creates compounds with very high melting points which conduct when molten or in solution but never when solid. Ionic compounds form what is known as a lattice structure. When these two charged particles come together they form an ionic bond because the positive magnesium ion is attracted to the negatively charged chloride ion. To become stable it must gain an electron to obtain a full outer energy level. Non-metals form negative ions because they gain electrons to become stable.Ĭhlorine (Cl) has an electron arrangement 2,8,7. They change into ions with a two positive charge. To become stable it must lose its two outer electrons to obtain a full outer energy level.Ītoms are neutral because they have equal numbers of protons and electrons however, when they lose two electrons they are no longer neutral. Magnesium (Mg) has the electron arrangement 2,8,2. 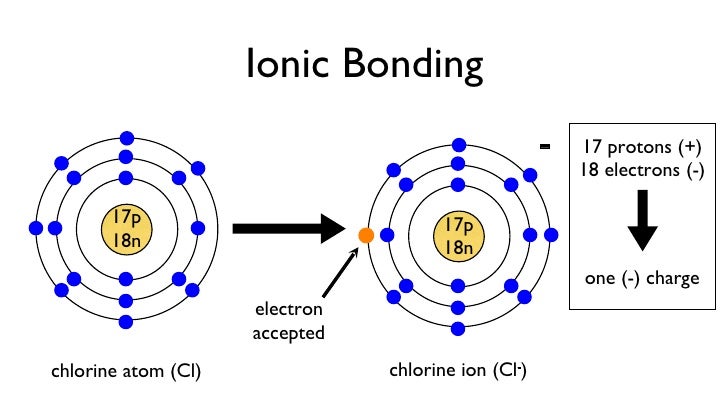
Metals form positive ions because they lose electrons to become stable. The ionic bond is the electrostatic force of attraction between a positively charged metal ion and a negatively charged non-metal ion. Ionic bonds are formed between a metal and non-metal, for example sodium chloride.Īn atom of sodium will lose an electron and form a positive ion.Īn atom of chlorine will gain an electron and form a negative ion. Learn about the basic structure of an ion, related to atomic number and mass.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
